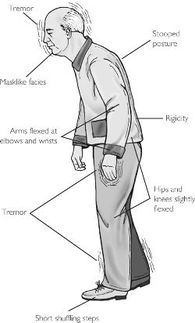
Research - Parkinson's Disease
Moxibustion Exerts a Neuroprotective Effect through Antiferroptosis in Parkinson’s Disease
Juan Lu,1 Xuelei Liu,1 Ye Tian,1 Hang Li,1 Zhenxing Ren,1 Shuang Liang,1 GuiyuZhang,1 Caiping Zhao,1 Xinrong Li,1 Tingting Wang,2 Dongfeng Chen,3Weihong Kuang,4 and Meiling Zhu
Abstract
The objective of this study was to explore the neuroprotective effect of moxibustion on rats with Parkinson’s disease (PD) and its mechanism. A Parkinson’s disease model was established in rats using a two-point stereotactic 6-hydroxydopamine injection in the right substantia nigra (SN) and ventral tegmental area. The rats received moxibustion at the Baihui (GV20) and Sishencong (EX-HN1) acupoints for 20 minutes, six times a week, for 6 weeks. The right SN tissue was histologically and immunohistochemically examined. Differentially expressed genes (DEGs) were identified through RNA sequencing. In addition, the levels of tyrosine hydroxylase (TH), glutathione peroxidase 4 (GPX4), and ferritin heavy chain 1 (FTH1) in SN were measured. In comparison to the model group, the moxibustion group showed a significantly greater TH immunoreactivity and a higher behavioural score. In particular, moxibustion led to an increase in the number and morphological stability of SN neural cells. The functional pathway analysis showed that DEGs are closely related to the ferroptosis pathway. GPX4 and FTH1 in the SN were significantly overexpressed in the moxibustion-treated rats with PD. Moxibustion can effectively reduce the death of SN neurons, decrease the occurrence of ferroptosis, and increase the TH activity to protect the neurons in rats with PD. The protective mechanism may be associated with suppression of the ferroptosis.
Source : Journal Evidence Based Complementary and Alternative Medicine
Link to Full Article
Estimated Residential Exposure to Agricultural Chemicals and Premature Mortality by Parkinson’s Disease in Washington State
Mariah Caballero 1,Solmaz Amiri 2,Justin T. Denney 3,Pablo Monsivais 2,Perry Hystad 4 andOfer Amram
Abstract
The aim of this study was to examine the relationship between estimated residential exposure to agricultural chemical application and premature mortality from Parkinson’s disease (PD) in Washington State. Washington State mortality records for 2011–2015 were geocoded using residential addresses, and classified as having exposure to agricultural land-use within 1000 meters. Generalized linear models were used to explore the association between land-use associated with agricultural chemical application and premature mortality from PD. Individuals exposed to land-use associated with glyphosate had 33% higher odds of premature mortality than those that were not exposed (Odds Ratio (OR) = 1.33, 95% Confidence Intervals (CI) = 1.06–1.67). Exposure to cropland associated with all pesticide application (OR = 1.19, 95% CI = 0.98–1.44) or Paraquat application (OR = 1.22, 95% CI = 0.99–1.51) was not significantly associated with premature mortality from PD, but the effect size was in the hypothesized direction. No significant associations were observed between exposure to Atrazine (OR = 1.21, 95% CI = 0.84–1.74) or Diazinon (OR = 1.07, 95% CI = 0.85–1.34), and premature mortality from PD. The relationship between pesticide exposure and premature mortality aligns with previous biological, toxicological, and epidemiological findings. Glyphosate, the world’s most heavily applied herbicide, and an active ingredient in Roundup® and Paraquat, a toxic herbicide, has shown to be associated with the odds of premature mortality from PD.
Source : Int. J. Environ. Res. Public Health
Link to Full Article
The Effect of Different Exercise Modes on Domain-Specific Cognitive Function in Patients Suffering from Parkinson’s Disease: A Systematic Review of Randomized Controlled Trials
Tim Stuckenschneidera,b,∗, Christopher D. Askewb, Annelise L. Meneses ˆ b, Ricarda Baakec , Jan Webera and Stefan Schneidera,b
Abstract
Background:Supervised exercise training alleviates motor symptoms in people with Parkinson’s disease (PD). However, the efficacy of exercise to improve nonmotor symptoms such as cognitive function is less well known.
Objective:To systematically review evidence on the efficacy of different exercise modes (coordination exercise, resistance exercise, aerobic exercise) on domain-specific cognitive function in patients with PD.
Methods:Parallel-group randomized controlled trials published before March 2018 were included. Primary outcome measures included global cognitive function and its subdomains, and the Unified Parkinson’s Disease Rating Scale was included as a secondary outcome. Methodological quality was assessed using the Physiotherapy Evidence Database scale.
Results:The literature search yielded 2,000 articles, of which 11 met inclusion criteria. 508 patients (mean age 68±4 years) were included with a disease severity from 1 to 4 on the Hoehn & Yahr stage scale. Overall study quality was modest (mean 6±2, range 3–8/10). In 5 trials a significant between-group effect size (ES) was identified for tests of specific cognitive domains, including a positive effect of aerobic exercise on memory (ES = 2.42) and executive function (ES = 1.54), and of combined resistance and coordination exercise on global cognitive function (ES = 1.54). Two trials found a significant ES for coordination exercise (ES = 0.84–1.88), which led to improved executive function compared with that of non-exercising control subjects.
Conclusion:All modes of exercise are associated with improved cognitive function in individuals with PD. Aerobic exercise tended to best improve memory; however, a clear effect of exercise mode was not identified.
Source : Journal Parkinsons Disease
Link to Full Article
Synergistic neuroprotection by coffee components eicosanoyl-5-hydroxytryptamide and caffeine in models of Parkinson's disease and DLB
Run Yan, Jie Zhang, Hye-Jin Park, Eun S. Park, Stephanie Oh, Haiyan Zheng, Eunsung Junn, Michael Voronkov, Jeffry B. Stock, and M. Maral Mouradian
Significance
Coffee consumption is linked with reduced risk of Parkinson’s disease (PD), and caffeine is generally believed to be the protective agent. However, several lines of evidence suggest the presence of additional compound(s) in coffee that can be protective as well. Here we show that eicosanoyl-5-hydroxytryptamide, which we purified from coffee as an agent that leads to enhanced enzymatic activity of the specific phosphatase PP2A that dephosphorylates the pathogenic protein α-synuclein, works in synergy with caffeine in protecting against mouse models of PD and Dementia with Lewy bodies. The mechanism of this synergy is also through enhancing PP2A, which is dysregulated in the brains of individuals with these α-synucleinopathies.
Abstract
Hyperphosphorylated α-synuclein in Lewy bodies and Lewy neurites is a characteristic neuropathological feature of Parkinson’s disease (PD) and Dementia with Lewy bodies (DLB). The catalytic subunit of the specific phosphatase, protein phosphatase 2A (PP2A) that dephosphorylates α-synuclein, is hypomethylated in these brains, thereby impeding the assembly of the active trimeric holoenzyme and reducing phosphatase activity. This phosphatase deficiency contributes to the accumulation of hyperphosphorylated α-synuclein, which tends to fibrillize more than unmodified α-synuclein. Eicosanoyl-5-hydroxytryptamide (EHT), a fatty acid derivative of serotonin found in coffee, inhibits the PP2A methylesterase so as to maintain PP2A in a highly active methylated state and mitigates the phenotype of α-synuclein transgenic (SynTg) mice. Considering epidemiologic and experimental evidence suggesting protective effects of caffeine in PD, we sought, in the present study, to test whether there is synergy between EHT and caffeine in models of α-synucleinopathy. Coadministration of these two compounds orally for 6 mo at doses that were individually ineffective in SynTg mice and in a striatal α-synuclein preformed fibril inoculation model resulted in reduced accumulation of phosphorylated α-synuclein, preserved neuronal integrity and function, diminished neuroinflammation, and improved behavioral performance. These indices were associated with increased levels of methylated PP2A in brain tissue. A similar profile of greater PP2A methylation and cytoprotection was found in SH-SY5Y cells cotreated with EHT and caffeine, but not with each compound alone. These findings suggest that these two components of coffee have synergistic effects in protecting the brain against α-synuclein−mediated toxicity through maintenance of PP2A in an active state.
Source : PNAS - Proceedings of the National Academy of Sciences
Link to Full Article
Can Exercise Help People with Parkinson’s Disease?
Exercise may help people with Parkinson’s disease improve their balance, ability to move around and quality of life, even if it does not reduce their risk of falling, according to a new study published in the December 31, 2014, online issue of Neurology®, the medical journal of the American Academy of Neurology.
For the study, 231 people with Parkinson’s disease either received their usual care or took part in an exercise program of 40 to 60 minutes of balance and leg strengthening exercises three times a week for six months. This minimally-supervised exercise program was prescribed and monitored by a physical therapist with participants performing most of the exercise at home. On average, 13 percent of the exercise sessions were supervised by a physical therapist.
Falling is a common problem for people with Parkinson’s, with 60 percent falling each year and two-thirds of those falling repeatedly. “The resulting injuries, pain, limitations of activity and fear of falling again can really affect people’s health and well-being,” said study author Colleen G. Canning, PhD, of the University of Sydney in Australia.
Compared to those in the control group, the number of falls by participants who exercised was reduced in those with less severe Parkinson’s disease, but not in those with more severe disease. For those with less severe disease a 70 percent reduction in falls was reported in those who exercised compared to those who did not.
“These results suggest that minimally supervised exercise programs aimed at reducing falls in people with Parkinson’s should be started early in the disease process,” Canning said.
Overall, those who took part in the exercise program performed better on tests of ability to move around and balance, had a lower fear of falls and reported better overall mood and quality of life.
Source : American Academy of Neurology
Link to Source
Finnish study establishes connection between gut microbiota and Parkinson’s disease
Parkinson’s disease sufferers have a different microbiota in their intestines than their healthy counterparts, according to a study conducted at the University of Helsinki and the Helsinki University Central Hospital. Researchers are now trying to determine what the connection between intestinal microbes and Parkinson’s disease is.
Our most important observation was that patients with Parkinson’s have much less bacteria from the Prevotellaceae family; unlike the control group, practically no one in the patient group had a large quantity of bacteria from this family, states DMSc Filip Scheperjans, neurologist at the Helsinki University Hospital (HUCH).
The researchers have not yet determined what the lack of Prevotellaceae bacteria in Parkinson's sufferers means – do these bacteria perhaps have a property which protects their host from the disease? Or does this discovery merely indicate that intestinal dysfunction is part of the pathology?
– It’s an interesting question which we are trying to answer, Sheperjans says.
Another interesting discovery was that the amount of bacteria from the Enterobacteriaceae family in the intestine was connected to the degree of severity of balance and walking problems in the patients. The more Enterobacteriaceae they had, the more severe the symptoms.
– We are currently re-examining these same subjects to determine whether the differences are permanent and whether intestinal bacteria are associated with the progression of the disease and therefore its prognosis,explains Sheperjans.
– In addition, we will have to see if these changes in the bacterial ecosystem are apparent before the onset of motor symptoms. We will of course also try to establish the basis of this connection between intestinal microbiota and Parkinson’s disease – what kind of mechanism binds them.
The researchers also hope that their discoveries could ultimately be used to develop a testing method which would improve the diagnostics in Parkinson’s disease and perhaps finally find a way to treat or even prevent Parkinson’s by focusing on gut microbiota.
Led by Scheperjans and Petri Auvinen from the University of Helsinki Institute of Biotechnology, the research project has received funding from the Michael J. Fox Foundation for Parkinson’s Research and the Finnish Parkinson Foundation.
The study recruited 72 Parkinson’s disease patients through HUCH and Hyvinkää Central Hospital, as well as an equal number of healthy control subjects.
Source : Helsinki University Central Hospital
Link to Source
Recent advances in herbal medicines treating Parkinson's disease
Abstract
Herbal medicines have attracted considerable attention in recent years, which are used to treat Parkinson's disease (PD) in China based on traditional Chinese medicine or modern pharmacological theories. We summarized and analyzed the anti-Parkinsonian activities of herbal medicines and herbal formulations investigated in PD models and provide future references for basic and clinical investigations. All the herbal medicines and herbal formulations were tested on PD models in vitro and in vivo. The relevant compounds and herbal extracts with anti-Parkinsonian activities were included and analyzed according to their genera or pharmacological activities. A total of 38 herbal medicines and 11 herbal formulations were analyzed. The relevant compounds, herbal extracts and formulations were reported to be effective on PD models by modulating multiple key events or signaling pathways implicated in the pathogenesis of PD. The plant species of these herbal medicines belong to 24 genera and 18 families, such as Acanthopanax, Alpinia and Astragalus, etc. These herbal medicines can be an alternative and valuable source for anti-Parkinsonian drug discovery. The plant species in these genera and families may be the most promising candidates for further investigation and deserve further consideration in clinical trials. Active components in some of the herbal extracts and the compatibility law of herbal formulations remain to be further investigated.
Source : Fitoterapia
Link to Full Article
Mucuna pruriens in Parkinson’s disease: a double blind clinical and pharmacological study
R Katzenschlager, A Evans, A Manson, P N Patsalos, N Ratnaraj, H Watt, L Timmermann,R Van der Giessen, A J Lees
Abstract
Background:The seed powder of the leguminous plant,Mucuna pruriens has long been used in traditional Ayurvedic Indian medicine for diseases including parkinsonism. We have assessed the clinical effects and levodopa (L-dopa) pharmacokinetics following two different doses of mucuna preparation and compared them with standard L-dopa/carbidopa (LD/CD).
Methods:Eight Parkinson’s disease patients with a short duration L-dopa response and on period dyskinesias completed a randomised, controlled, double blind crossover trial. Patients were challenged with single doses of 200/50 mg LD/CD, and 15 and 30 g of mucuna preparation in randomised order at weekly intervals.L-Dopa pharmacokinetics were determined, and Unified Parkinson’s Disease Rating Scale and tapping speed were obtained at baseline and repeatedly during the 4 h following drug ingestion. Dyskinesias were assessed using modified
AIMS and Goetz scales.Results:Compared with standard LD/CD, the 30 g mucuna preparation led to a considerably faster on set of effect (34.6v68.5 min; p=0.021), reflected in shorter latencies to peak L-dopa plasma concentrations.Mean on time was 21.9% (37 min) longer with 30 g mucuna than with LD/CD (p=0.021); peak L-dopaplasma concentrations were 110% higher and the area under the plasma concentration vtime curve (area under curve) was 165.3% larger (p=0.012). No significant differences in dyskinesias or tolerability occurred.
Conclusions:The rapid onset of action and longer on time without concomitant increase in dyskinesias on mucuna seed powder formulation suggest that this natural source of L-dopa might possess advantages over conventional L-dopa preparations in the long term management of PD. Assessment of long term efficacy and tolerability in a randomised, controlled study is warranted
Source : J Neurol Neurosurg Psychiatry
Link to Full Article
Identification of Licopyranocoumarin and Glycyrurol from Herbal Medicines as Neuroprotective Compounds for Parkinson's Disease
Takahiro Fujimaki, Shinji Saiki, Etsu Tashiro, Daisuke Yamada, Mitsuhiro Kitagawa, Nobutaka Hattori mail, Masaya Imoto mail
Abstract
In the course of screening for the anti-Parkinsonian drugs from a library of traditional herbal medicines, we found that the extracts of choi-joki-to and daio-kanzo-to protected cells from MPP+-induced cell death. Because choi-joki-to and daio-kanzo-to commonly contain the genus Glycyrrhiza, we isolated licopyranocoumarin (LPC) and glycyrurol (GCR) as potent neuroprotective principals from Glycyrrhiza. LPC and GCR markedly blocked MPP+-induced neuronal PC12D cell death and disappearance of mitochondrial membrane potential, which were mediated by JNK. LPC and GCR inhibited MPP+-induced JNK activation through the suppression of reactive oxygen species (ROS) generation, thereby inhibiting MPP+-induced neuronal PC12D cell death. These results indicated that LPC and GCR derived from choi-joki-to and daio-kanzo-to would be promising drug leads for PD treatment in the future.
Source : PlosOne
Link to Full Article
Neuroprotective Effects of Jitai Tablet, a Traditional Chinese Medicine, on the MPTP-Induced Acute Model of Parkinson’s Disease: Involvement of the Dopamine System
Jia Liu,1 Jinlong Gao,1 Shaoang Tu,1 Shasha Xu,1 Ying Liu,1 Weihu Shang,1 Chenxin Gu,1 Yiyun Huang,2 and Mei Han1,3
1College of Chemistry, Beijing Normal University, Beijing 100875, China
2Yale PET Center, Department of Diagnostic Radiology, Yale University School of Medicine, New Haven, CT 06510, USA
3Key Laboratory of Radiopharmaceuticals, Ministry of Education, College of Chemistry, Beijing Normal University, Beijing 100875, China
Abstract
Jitai tablet (JTT) is a traditional Chinese medicine used to treat neuropsychiatric disorders. We previously demonstrated that JTT treatment led to increased level of dopamine transporter (DAT) in the striatum, thus indicating that JTT might have therapeutic potential for Parkinson’s disease (PD), which is characterized by dysregulated dopamine (DA) transmission and decreased striatal DAT expression. The aim of this study was to investigate the neuroprotective effect of JTT on MPTP-induced PD mice. Using locomotor activity test and rotarod test, we evaluated the effects of JTT (0.50, 0.15, or 0.05 g/kg) on MPTP-induced behavioral impairments. Tyrosine hydroxylase TH-positive neurons in the substantia nigra and DAT and dopamine D2 receptor (D2R) levels in the striatum were detected by immunohistochemical staining and/or autoradiography. Levels of DA and its metabolites were determined by HPLC. In MPTP-treated mice, behavioral impairments were alleviated by JTT treatment. Moreover, JTT protected against impairment of TH-positive neurons and attenuated the MPTP-induced decreases in DAT and D2R. Finally, high dose of JTT (0.50 g/kg) inhibited the MPTP-induced increase in DA metabolism rate. Taken together, results from our present study provide evidence that JTT offers neuroprotective effects against the neurotoxicity of MPTP and thus might be a potential treatment for PD.
Source : Journal Evidence Based Complementary and Alternative Medicine,
Link to Full Article
Resveratrol Partially Prevents Rotenone-Induced Neurotoxicity in Dopaminergic SH-SY5Y Cells through Induction of Heme Oxygenase-1 Dependent Autophagy
by Tsu-Kung Lin, Shang-Der Chen, Yao-Chung Chuang, Hung-Yu Lin, Chi-Ren Huang, Jiin-Haur Chuang, Pei-Wen Wang, Sheng-Teng Huang, Mao-Meng Tiao, Jin-Bor Chen and Chia-Wei Liou
Abstract:
Parkinson disease (PD) is a complex neurodegenerative disorder characterized by a progressive loss of dopaminergic neurons. Mitochondrial dysfunction, oxidative stress or protein misfolding and aggregation may underlie this process. Autophagy is an intracellular catabolic mechanism responsible for protein degradation and recycling of damaged proteins and cytoplasmic organelles. Autophagic dysfunction may hasten the progression of neuronal degeneration. In this study, resveratrol promoted autophagic flux and protected dopaminergic neurons against rotenone-induced apoptosis. In an in vivo PD model, rotenone induced loss of dopaminergic neurons, increased oxidation of mitochondrial proteins and promoted autophagic vesicle development in brain tissue. The natural phytoalexin resveratrol prevented rotenone-induced neuronal apoptosis in vitro, and this pro-survival effect was abolished by an autophagic inhibitor. Although both rotenone and resveratrol promoted LC3-II accumulation, autophagic flux was inhibited by rotenone and augmented by resveratrol. Further, rotenone reduced heme oxygenase-1 (HO-1) expression, whereas resveratrol increased HO-1 expression. Pharmacological inhibition of HO-1 abolished resveratrol-mediated autophagy and neuroprotection. Notably, the effects of a pharmacological inducer of HO-1 were similar to those of resveratrol, and protected against rotenone-induced cell death in an autophagy-dependent manner, validating the hypothesis of HO-1 dependent autophagy in preventing neuronal death in the in vitro PD model. Collectively, our findings suggest that resveratrol induces HO-1 expression and prevents dopaminergic cell death by regulating autophagic flux; thus protecting against rotenone-induced neuronal apoptosis.
Source : Int. J. Mol. Sci. 2014
Link to download Full Article
Study Shows Exercise Improves Depression in Parkinson's Patients
A new study by a movement disorder neurologist at The Ohio State University Wexner Medical Center found that depression improved among patients with Parkinson's disease who participated in a long-term group exercise program.
The findings are published in the journal Parkinsonism and Related Disorders.
Parkinson's disease is a progressive neurodegenerative disorder that affects more than 1 million Americans. Each year, about 60,000 Americans are newly diagnosed with Parkinson's disease. Parkinson's patients develop progressive disability over time, despite the best pharmacological and surgical management. Therefore, complementary approaches that can maximize functional ability and improve quality of life are important.
In this pilot study, 31 patients with Parkinson's disease were randomly assigned to an "early start group" or a "delayed start group" for a rigorous formal group exercise program that met for one hour, three days a week. The early start group exercised for a total of 48 weeks, while the delayed start group exercised only the last 24 weeks. One patient dropped out of the study.
"Our findings demonstrate that long-term group exercise programs are feasible in the Parkinson's disease population. Patients enjoyed exercising, and they stayed with the program that included cardiovascular and resistance training," said principal investigator Dr. Ariane Park, a movement disorder neurologist at the Madden Center for Parkinson's Disease and Related Disorders at Ohio State's Wexner Medical Center.
"Our study also showed that earlier participation in a group exercise program significantly improved symptoms of depression compared to the delayed start group, and this is important because often mood can be more debilitating than motor symptoms," Park said.
More than 50 percent of patients with Parkinson's disease suffer from depression, which is why it is important to help patients find new ways to cope and improve their symptoms, said Park, who also is a researcher in Ohio State's Neuroscience Program.
Researchers had also hoped to show that exercise could slow the progression of the disease, but the study did not provide strong evidence of any 'neuroprotective' effect on motor function, possibly because of the small sample size, Park said.
"We recommend exercise to all of our Parkinson's patients. Currently, there is no consensus on a standardized physical exercise regimen with regard to type, frequency and intensity. The literature supports that any routine that improves physical fitness is good for Parkinson's disease -- and that can include walking, swimming, tai chi or even dancing," Park said. "We just want patients to move on a regular basis. Not only will they move better, but they will feel better."
Source Science Daily:
Link to Source
Unrecognized vitamin D3 deficiency is common in Parkinson disease
Harvard Biomarker Study
Abstract
Objective: To conclusively test for a specific association between the biological marker 25-hydroxy-vitamin D3, a transcriptionally active hormone produced in human skin and liver, and the prevalence and severity of Parkinson disease (PD).
Methods: We used liquid chromatography/tandem mass spectrometry to establish an association specifically between deficiency of 25-hydroxy-vitamin D3 and PD in a cross-sectional and longitudinal case-control study of 388 patients (mean Hoehn and Yahr stage of 2.1 ± 0.6) and 283 control subjects free of neurologic disease nested in the Harvard Biomarker Study.
Results: Plasma levels of 25-hydroxy-vitamin D3 were associated with PD in both univariate and multivariate analyses with p values = 0.0034 and 0.047, respectively. Total 25-hydroxy-vitamin D levels, the traditional composite measure of endogenous and exogenous vitamin D, were deficient in 17.6% of patients with PD compared with 9.3% of controls. Low 25-hydroxy-vitamin D3 as well as total 25-hydroxy-vitamin D levels were correlated with higher total Unified Parkinson’s Disease Rating Scale scores at baseline and during follow-up.
Conclusions: Our study reveals an association between 25-hydroxy-vitamin D3 and PD and suggests that thousands of patients with PD in North America alone may be vitamin D–deficient. This finding has immediate relevance for individual patients at risk of falls as well as public health, and warrants further investigation into the mechanism underlying this association.
Source : Journal Neurology
Link to Abstract
A Cup of Joe May Help Some Parkinson’s Disease Symptoms
While drinking caffeine each day does not appear to help improve sleepiness among people with Parkinson’s disease, it may have a benefit in controlling movement, according to new research published in the August 1, 2012, online issue of Neurology®, the medical journal of the American Academy of Neurology. “Studies have shown that people who use caffeine are less likely to develop Parkinson’s disease, but this is one of the first studies in humans to show that caffeine can help with movement symptoms for people who already have the disease,” said study author Ronald Postuma, MD, MSc, with McGill University in Montreal and the Research Institute of the McGill University Health Center. Postuma is also a member of the American Academy of Neurology.
For the study, 61 people with Parkinson’s disease who showed symptoms of daytime sleepiness and some motor symptoms were given either a placebo pill or a pill with 100 milligrams of caffeine two times a day for three weeks, then 200 milligrams twice a day for three weeks, which was the equivalent of between two and four cups of coffee per day.
After six weeks, the half that took the caffeine supplements averaged a five-point improvement in Parkinson’s severity ratings compared to those who didn’t consume caffeine. “This is a modest improvement, but may be enough to provide benefit to patients. On the other hand, it may not be sufficient to explain the relationship between caffeine non-use and Parkinson's, since studies of the progression of Parkinson’s symptoms early in the disease suggest that a five-point reduction would delay diagnosis by only six months,” said Postuma.
The caffeine group also averaged a three-point improvement in the speed of movement and amount of stiffness compared to the placebo group. Caffeine did not appear to help improve daytime sleepiness and there were no changes in quality of life, depression or sleep quality in study participants.
“The study is especially interesting since caffeine seems to block a malfunctioning brain signal in Parkinson’s disease and is so safe and inexpensive,” said Michael Schwarzschild, MD, PhD, of Massachusetts General Hospital in Boston, who wrote an accompanying editorial. “Although the results do not suggest that caffeine should be used as a treatment in Parkinson’s disease, they can be taken into consideration when people with Parkinson’s are discussing their caffeine use with their neurologist.” Schwarzschild is also a member of the American Academy of Neurology.
The study authors noted that the length of the study was short and that the effects of caffeine may lessen over time.
Source : Newswise
Link to Source
Parkinson’s Disease: An Integrative Approach - A Natural Standard Monograph
Catherine Ulbricht, PharmD,
| laura_-_parkinsons_disease_-_an_integrative_approach.pdf | |
| File Size: | 678 kb |
| File Type: | |
_Curcumin nudges sluggish protein folding
A compound in the spice turmeric may be an effective way to prevent protein clumping—the first step in Parkinson’s disease
Researchers led by Basir Ahmad, a postdoctoral researcher at Michigan State University, demonstrated earlier this year that slow-wriggling alpha-synuclein proteins are the cause of the clumping, or aggregation.
A new study published in the Journal of Biological Chemistry, shows that curcumin can prevent it.
“Our research shows that curcumin can rescue proteins from aggregation, the first steps of many debilitating diseases,” says Lisa Lapidus, associate professor of physics and astronomy at MSU who co-authored the paper with Ahmad.
“More specifically, curcumin binds strongly to alpha-synuclein and prevents aggregation at body temperatures.”
Lapidus’ lab uses lasers to study protein folding. Proteins are chains of amino acids that do most of the work in cells. Scientists understand protein structure, but they don’t know how they are built—a process known as folding.
The new research sheds light on the process by correlating the speed at which protein folds with its tendency to clump or bind with other proteins.
When curcumin attaches to alpha-synuclein it not only stops clumping, but it also raises the protein’s folding or reconfiguration rate. By bumping up the speed, curcumin moves the protein out of a dangerous speed zone allowing it to avoid clumping with other proteins.
Finding a compound that can fix a protein when it first begins to misfold can lead scientists to identify drugs that can treat certain diseases. But doctors won’t be prescribing curcumin pills any time soon, Lapidus says.
“Curcumin’s usefulness as an actual drug may be pretty limited since it doesn’t go into the brain easily where this misfolding is taking place,” she says.
“But this kind of study showcases the technique of measuring reconfiguration and opens the door for developing drug treatments.”
Source : Futurity
Link to Source
Discovering new causes of Parkinson’s
A new study suggests that exposure to the common industrial solvent TCE or trichloroethylene may lead to Parkinson’s disease. Host Steve Curwood speaks with Dr. Samuel Goldman of The Parkinson’s Institute in California about the disease and how his team identified potential environmental causes.
Transcript CURWOOD: Parkinson’s disease afflicts some 700,000 Americans. The disorder often causes tremors and an unsteady gait. And more than many other diseases, Parkinson’s is linked to environmental exposures. Now comes news that among the possible causes of the disease is a common workplace chemical. Joining me to talk with about the new research is Dr. Samuel Goldman, one of the authors. Welcome, Sir.
GOLDMAN: Thank you. Thanks for having me.
CURWOOD: My pleasure. Your study is really interesting. You looked at twins, and you looked at twins where one brother has Parkinson’s disease and one doesn’t. And you then reconstructed a detailed work history for them - any place they’d worked for more than six months since they were 10-years-old. What did you find?
GOLDMAN: We found that the twin who was exposed to a compound called trichloroethylene, or TCE, had a more than six-fold increased risk of having Parkinson’s disease than their unexposed co-twin. And there were also some other solvents that we looked at as well, including one called PERC, or perchloroethylene, which is the most common dry cleaning solvent, and that was also associated with a markedly increased risk of Parkinsons.
CURWOOD: What if they had been exposed to TCE or PERC?
GOLDMAN: Exposure to TCE or PERC was associated with a nearly nine-fold increased risk of Parkinson’s disease.
CURWOOD: That’s a startling number, isn’t it? You must have been pretty excited to see this.
GOLDMAN: Well, excited, dismayed… We’ve always believed that the vast majority of Parkinson’s disease is a consequence of environmental risk factors, but it’s important to recognize that this is a single study, so replication of our results is really important at this point before we can progress to being really certain that this is a causal link.
CURWOOD: Talk to me a bit about these chemicals. TCE, now that’s trichloroethylene, now what does it do?
GOLDMAN: TCE was extremely widely used - it still is, but not so much as 30 years ago. So it would be used, now, primarily for degreasing of metal parts in manufacturing. And in fact, it’s in a broad range of consumer products and it has been for decades. And actually, up and through the 1970s, TCE was used to de-caffeinate coffee. It has been used as a general anesthetic. It was in wide use during the 1940s and 1950s primarily as an obstetrical anesthetic, strangely.
CURWOOD: So, tell me a bit about how you did design this study. You got almost a hundred sets of twins?
GOLDMAN: Correct. Well, we’ve been following this cohort of twins since the early 90s, and then that’s a very powerful study design to be able to look at environmental risk associations with disease.
CURWOOD: Why?
GOLDMAN: Because twins, if they’re identical twins, they are genetically identical. And if they are fraternal twins, they are at least genetically very similar. So we are able to remove the genetic background effect from the equation and focus specifically on differences in environmental exposures.
CURWOOD: Now, what were the most occupations for the people who got sick?
GOLDMAN: The most common occupational exposure settings for TCE and PERC were among electricians and dry cleaners as well as people who repaired industrial machinery and artists.
CURWOOD: Artists?
GOLDMAN: Artists use solvents commonly.
CURWOOD: Of course art is a career; it’s also a hobby. What hobbies were people most at risk of getting Parkinson’s from this toxic exposure?
GOLDMAN: The hobbies that we observed were people working in carpentry. Artists again, people who worked in photography in particular. But oftentimes it’s in the household settings where exposure levels can be exceedingly high because there is no one enforcing a regulatory maximum or protective equipment in the home.
CURWOOD: So, Dr. Goldman, if somebody came into your office today and said, ‘Gee, I read your study. I want you to know that I worked at an aircraft engine repair place and was up to my elbows everyday in TCE. I’m fine today.’ What would you tell him he could do to help keep from getting Parkinson’s?
GOLDMAN: I would love to be able to offer some advice, but currently there really is no way that we’re aware of, to delay or prevent Parkinson’s disease. I think the most important thing we can do right now is to replicate this observation and if it’s found to be a consistently observed link between exposure to these compounds and Parkinson’s, I would hope that the funding agencies would really get behind this work and help us move it forward.
CURWOOD: And what are we to make of the fact that so many environmental contaminants seem to be linked, or even perhaps cause, Parkinson’s disease - there are earlier studies that have shown that have shown that several pesticides cause it by destroying brain cells, and research also points to, what, some heavy metals like manganese?
GOLDMAN: To me, as an investigator, for many years in Parkinson’s disease, I am really shocked at the paucity of environmental factors that we’ve been able to identify.
CURWOOD: Paucity? Too few?
GOLDMAN: I would like to know what causes Parkinson’s disease. So I feel like we’ve really only begun to scratch the surface of identifying risk factors for Parkinson’s disease. What’s really interesting about Parkinson’s is that there are very few naturally occurring disease clusters.
When you find a cluster, it’s a good sign that there’s likely to be a shared environmental determinant in those people. But there are very few naturally occurring clusters of Parkinson’s disease. So that implies that the environmental factors that go into causing Parkinson’s disease are likely spread out over a very long period of time and maybe different in everyone.
So what we’ve been able to link so far is, as you pointed out, several pesticides have been linked, but only a couple - paraquat - an herbicide -, and rotenone - an insecticide. As someone who has worked in this field for many years, I’m somewhat discouraged at the small number of environmental compounds that have been definitively linked with Parkinson’s disease.
CURWOOD: What you’re saying to me is that Parkinson’s disease as a disorder might be akin to, if you’ll forgive me, a broken leg. In other words, you can break your leg skiing, it could be in a car crash, you could fall over in your garden, you could get hit by a door. That it’s the endpoint of any number of processes.
GOLDMAN: That’s absolutely right. I think that we’ll find that there are many environmental insults that ultimately coalesce to result in Parkinson’s disease. But that in any given individual, the route to get to that point is different.
CURWOOD: Sam Goldman, thanks so much for talking with us today.
GOLDMAN: Well, it’s been a real pleasure and thanks for your interest in our work.
CURWOOD: Sam Goldman is a physician and public health researcher with the Parkinson’s Institute in Sunnyvale, California. He was part of a team whose research appears in the Current issue of the Annals of Neurology.
Source : Living on Earth
Link to Source + Listen to Interview
Vitamin D deficiency linked to Parkinson's disease, cognitive decline
Studies of vitamin D have been on the rise in recent years, and with good reason--a 2009 estimate suggests that nearly three quarters of teens and adults in the U.S. are deficient in this vital nutrient. Vitamin D deficiency not only causes rickets, a skeletal disorder in which the bones are soft and weak, but has also been associated with a rapidly increasing range of chronic conditions like cancer, heart disease, and type 2 diabetes. Now, two new studies suggest a link between vitamin D and neurological disorder: Older people with insufficient vitamin D levels may be more likely to develop Parkinson's disease and experience cognitive decline. The first, led by Paul Knekt and colleagues at the National Institute for Health and Welfare, Finland, examined levels of vitamin D in the blood of 3,173 Finnish men and women aged 50 to 79 determined to be free of Parkinson's disease at the start of the study. The researchers then examined the incidence of Parkinson's disease in these participants over a 29-year follow-up period. They found that participants with the highest levels of vitamin D (more than 50 nmol/L) had a 65 percent lower risk of developing Parkinson's disease than those with the lowest vitamin D levels (less than 25 nmol/L). The researchers accounted for potentially confounding variables such as age, sex, marital status, education, alcohol consumption, smoking, physical activity and month of blood draw.
Parkinson's disease is a progressive neurodegenerative disorder that leads to impaired movement and speech, and is thought to result from insufficient dopamine levels in the brain. How vitamin D may protect against Parkinson's is not understood, although there is limited evidence from cell-based and animal models that vitamin D may prevent the loss of dopaminergic neurons (cells that produce dopamine).
One important limitation to the study is that the average vitamin D concentration of all the study participants (approximately 40 nmol/L) falls well below what is considered to be optimal (more than 75 nmol/L). Therefore, whether supplementation with vitamin D would further lower the risk for Parkinson's remains unknown. Nevertheless, the study suggests that not having enough vitamin D may predispose individuals to Parkinson's, and provides a starting point for further investigation. The results were published online July 12 in the Archives of Neurology.
In the second study, David Llewellyn of the University of Exeter and colleagues examined vitamin D levels among 858 Italian men and women age 65 and older. They found that more than half of the participants with dementia were vitamin D deficient (less than 50 nmol/L). What's more, cognitive tests revealed that severely deficient individuals (less than 25 nmol/L) were 60 percent more likely to undergo cognitive decline over the six-year follow-up period. This study appears online July 12 in the Archives of Internal Medicine.
Humans can obtain vitamin D by eating oily fish or fortified foods, and it is also photosynthesized in the skin upon exposure to adequate amounts of ultraviolet B (UVB) rays in sunlight. Major factors that influence vitamin D status in humans include season, latitude, age, skin tone, diet and supplement use. The U.S. Institute of Medicine currently recommends that adult men and women aim for a daily intake of 200 to 600 International Units (IU) of vitamin D.
New guidelines for vitamin D intake were published online July 12 in the Canadian Medical Association Journal by scientists from Osteoporosis Canada, a nonprofit organization. Because vitamin D influences calcium absorption and may protect against osteoporosis, the authors advise an increased daily intake of 400 to 1000 IU for healthy Canadians under age 50, and up to 2000 IU for those older than 50. The researchers state the changes are necessary because winter sunlight north of the 35th parallel (which coincides with the southern border of Tennessee) provides insufficient UVB for people living in that region to adequately make vitamin D.
The studies by Knekt and Llewellyn are not the first to link vitamin D deficiency with neurological problems, however. A role for vitamin D has previously been suggested in multiple sclerosis, autism and schizophrenia.
Some experts advise interpreting the results of these and other observational studies of vitamin D with caution. The above studies relied on participants from specific geographic areas, so more study is needed to determine whether the findings apply to other regions. Furthermore, "low vitamin D levels may simply be a marker for lower health status rather than a cause of it," Andrew Grey, professor of medicine at the University of Auckland, wrote in an editorial in the Archives of Internal Medicine. This is because vitamin D levels are directly related to sunlight exposure and physical activity; less healthy individuals are therefore likely to be less active and more sunlight-deprived, and have lower levels of vitamin D.
"It is now time to test the various hypotheses generated by observational studies of vitamin D…in adequately designed and conducted randomized controlled trials," Grey concluded. "We should invest in trials that provide the best possible evidence on the benefits and risks of vitamin D before we invest in costly, difficult, and potentially unrewarding interventional strategies."
Source: Scientific America 15/7/2010
LINK TO SOURCE
_
A compound in the spice turmeric may be an effective way to prevent protein clumping—the first step in Parkinson’s disease
Researchers led by Basir Ahmad, a postdoctoral researcher at Michigan State University, demonstrated earlier this year that slow-wriggling alpha-synuclein proteins are the cause of the clumping, or aggregation.
A new study published in the Journal of Biological Chemistry, shows that curcumin can prevent it.
“Our research shows that curcumin can rescue proteins from aggregation, the first steps of many debilitating diseases,” says Lisa Lapidus, associate professor of physics and astronomy at MSU who co-authored the paper with Ahmad.
“More specifically, curcumin binds strongly to alpha-synuclein and prevents aggregation at body temperatures.”
Lapidus’ lab uses lasers to study protein folding. Proteins are chains of amino acids that do most of the work in cells. Scientists understand protein structure, but they don’t know how they are built—a process known as folding.
The new research sheds light on the process by correlating the speed at which protein folds with its tendency to clump or bind with other proteins.
When curcumin attaches to alpha-synuclein it not only stops clumping, but it also raises the protein’s folding or reconfiguration rate. By bumping up the speed, curcumin moves the protein out of a dangerous speed zone allowing it to avoid clumping with other proteins.
Finding a compound that can fix a protein when it first begins to misfold can lead scientists to identify drugs that can treat certain diseases. But doctors won’t be prescribing curcumin pills any time soon, Lapidus says.
“Curcumin’s usefulness as an actual drug may be pretty limited since it doesn’t go into the brain easily where this misfolding is taking place,” she says.
“But this kind of study showcases the technique of measuring reconfiguration and opens the door for developing drug treatments.”
Source : Futurity
Link to Source
Discovering new causes of Parkinson’s
A new study suggests that exposure to the common industrial solvent TCE or trichloroethylene may lead to Parkinson’s disease. Host Steve Curwood speaks with Dr. Samuel Goldman of The Parkinson’s Institute in California about the disease and how his team identified potential environmental causes.
Transcript CURWOOD: Parkinson’s disease afflicts some 700,000 Americans. The disorder often causes tremors and an unsteady gait. And more than many other diseases, Parkinson’s is linked to environmental exposures. Now comes news that among the possible causes of the disease is a common workplace chemical. Joining me to talk with about the new research is Dr. Samuel Goldman, one of the authors. Welcome, Sir.
GOLDMAN: Thank you. Thanks for having me.
CURWOOD: My pleasure. Your study is really interesting. You looked at twins, and you looked at twins where one brother has Parkinson’s disease and one doesn’t. And you then reconstructed a detailed work history for them - any place they’d worked for more than six months since they were 10-years-old. What did you find?
GOLDMAN: We found that the twin who was exposed to a compound called trichloroethylene, or TCE, had a more than six-fold increased risk of having Parkinson’s disease than their unexposed co-twin. And there were also some other solvents that we looked at as well, including one called PERC, or perchloroethylene, which is the most common dry cleaning solvent, and that was also associated with a markedly increased risk of Parkinsons.
CURWOOD: What if they had been exposed to TCE or PERC?
GOLDMAN: Exposure to TCE or PERC was associated with a nearly nine-fold increased risk of Parkinson’s disease.
CURWOOD: That’s a startling number, isn’t it? You must have been pretty excited to see this.
GOLDMAN: Well, excited, dismayed… We’ve always believed that the vast majority of Parkinson’s disease is a consequence of environmental risk factors, but it’s important to recognize that this is a single study, so replication of our results is really important at this point before we can progress to being really certain that this is a causal link.
CURWOOD: Talk to me a bit about these chemicals. TCE, now that’s trichloroethylene, now what does it do?
GOLDMAN: TCE was extremely widely used - it still is, but not so much as 30 years ago. So it would be used, now, primarily for degreasing of metal parts in manufacturing. And in fact, it’s in a broad range of consumer products and it has been for decades. And actually, up and through the 1970s, TCE was used to de-caffeinate coffee. It has been used as a general anesthetic. It was in wide use during the 1940s and 1950s primarily as an obstetrical anesthetic, strangely.
CURWOOD: So, tell me a bit about how you did design this study. You got almost a hundred sets of twins?
GOLDMAN: Correct. Well, we’ve been following this cohort of twins since the early 90s, and then that’s a very powerful study design to be able to look at environmental risk associations with disease.
CURWOOD: Why?
GOLDMAN: Because twins, if they’re identical twins, they are genetically identical. And if they are fraternal twins, they are at least genetically very similar. So we are able to remove the genetic background effect from the equation and focus specifically on differences in environmental exposures.
CURWOOD: Now, what were the most occupations for the people who got sick?
GOLDMAN: The most common occupational exposure settings for TCE and PERC were among electricians and dry cleaners as well as people who repaired industrial machinery and artists.
CURWOOD: Artists?
GOLDMAN: Artists use solvents commonly.
CURWOOD: Of course art is a career; it’s also a hobby. What hobbies were people most at risk of getting Parkinson’s from this toxic exposure?
GOLDMAN: The hobbies that we observed were people working in carpentry. Artists again, people who worked in photography in particular. But oftentimes it’s in the household settings where exposure levels can be exceedingly high because there is no one enforcing a regulatory maximum or protective equipment in the home.
CURWOOD: So, Dr. Goldman, if somebody came into your office today and said, ‘Gee, I read your study. I want you to know that I worked at an aircraft engine repair place and was up to my elbows everyday in TCE. I’m fine today.’ What would you tell him he could do to help keep from getting Parkinson’s?
GOLDMAN: I would love to be able to offer some advice, but currently there really is no way that we’re aware of, to delay or prevent Parkinson’s disease. I think the most important thing we can do right now is to replicate this observation and if it’s found to be a consistently observed link between exposure to these compounds and Parkinson’s, I would hope that the funding agencies would really get behind this work and help us move it forward.
CURWOOD: And what are we to make of the fact that so many environmental contaminants seem to be linked, or even perhaps cause, Parkinson’s disease - there are earlier studies that have shown that have shown that several pesticides cause it by destroying brain cells, and research also points to, what, some heavy metals like manganese?
GOLDMAN: To me, as an investigator, for many years in Parkinson’s disease, I am really shocked at the paucity of environmental factors that we’ve been able to identify.
CURWOOD: Paucity? Too few?
GOLDMAN: I would like to know what causes Parkinson’s disease. So I feel like we’ve really only begun to scratch the surface of identifying risk factors for Parkinson’s disease. What’s really interesting about Parkinson’s is that there are very few naturally occurring disease clusters.
When you find a cluster, it’s a good sign that there’s likely to be a shared environmental determinant in those people. But there are very few naturally occurring clusters of Parkinson’s disease. So that implies that the environmental factors that go into causing Parkinson’s disease are likely spread out over a very long period of time and maybe different in everyone.
So what we’ve been able to link so far is, as you pointed out, several pesticides have been linked, but only a couple - paraquat - an herbicide -, and rotenone - an insecticide. As someone who has worked in this field for many years, I’m somewhat discouraged at the small number of environmental compounds that have been definitively linked with Parkinson’s disease.
CURWOOD: What you’re saying to me is that Parkinson’s disease as a disorder might be akin to, if you’ll forgive me, a broken leg. In other words, you can break your leg skiing, it could be in a car crash, you could fall over in your garden, you could get hit by a door. That it’s the endpoint of any number of processes.
GOLDMAN: That’s absolutely right. I think that we’ll find that there are many environmental insults that ultimately coalesce to result in Parkinson’s disease. But that in any given individual, the route to get to that point is different.
CURWOOD: Sam Goldman, thanks so much for talking with us today.
GOLDMAN: Well, it’s been a real pleasure and thanks for your interest in our work.
CURWOOD: Sam Goldman is a physician and public health researcher with the Parkinson’s Institute in Sunnyvale, California. He was part of a team whose research appears in the Current issue of the Annals of Neurology.
Source : Living on Earth
Link to Source + Listen to Interview
Vitamin D deficiency linked to Parkinson's disease, cognitive decline
Studies of vitamin D have been on the rise in recent years, and with good reason--a 2009 estimate suggests that nearly three quarters of teens and adults in the U.S. are deficient in this vital nutrient. Vitamin D deficiency not only causes rickets, a skeletal disorder in which the bones are soft and weak, but has also been associated with a rapidly increasing range of chronic conditions like cancer, heart disease, and type 2 diabetes. Now, two new studies suggest a link between vitamin D and neurological disorder: Older people with insufficient vitamin D levels may be more likely to develop Parkinson's disease and experience cognitive decline. The first, led by Paul Knekt and colleagues at the National Institute for Health and Welfare, Finland, examined levels of vitamin D in the blood of 3,173 Finnish men and women aged 50 to 79 determined to be free of Parkinson's disease at the start of the study. The researchers then examined the incidence of Parkinson's disease in these participants over a 29-year follow-up period. They found that participants with the highest levels of vitamin D (more than 50 nmol/L) had a 65 percent lower risk of developing Parkinson's disease than those with the lowest vitamin D levels (less than 25 nmol/L). The researchers accounted for potentially confounding variables such as age, sex, marital status, education, alcohol consumption, smoking, physical activity and month of blood draw.
Parkinson's disease is a progressive neurodegenerative disorder that leads to impaired movement and speech, and is thought to result from insufficient dopamine levels in the brain. How vitamin D may protect against Parkinson's is not understood, although there is limited evidence from cell-based and animal models that vitamin D may prevent the loss of dopaminergic neurons (cells that produce dopamine).
One important limitation to the study is that the average vitamin D concentration of all the study participants (approximately 40 nmol/L) falls well below what is considered to be optimal (more than 75 nmol/L). Therefore, whether supplementation with vitamin D would further lower the risk for Parkinson's remains unknown. Nevertheless, the study suggests that not having enough vitamin D may predispose individuals to Parkinson's, and provides a starting point for further investigation. The results were published online July 12 in the Archives of Neurology.
In the second study, David Llewellyn of the University of Exeter and colleagues examined vitamin D levels among 858 Italian men and women age 65 and older. They found that more than half of the participants with dementia were vitamin D deficient (less than 50 nmol/L). What's more, cognitive tests revealed that severely deficient individuals (less than 25 nmol/L) were 60 percent more likely to undergo cognitive decline over the six-year follow-up period. This study appears online July 12 in the Archives of Internal Medicine.
Humans can obtain vitamin D by eating oily fish or fortified foods, and it is also photosynthesized in the skin upon exposure to adequate amounts of ultraviolet B (UVB) rays in sunlight. Major factors that influence vitamin D status in humans include season, latitude, age, skin tone, diet and supplement use. The U.S. Institute of Medicine currently recommends that adult men and women aim for a daily intake of 200 to 600 International Units (IU) of vitamin D.
New guidelines for vitamin D intake were published online July 12 in the Canadian Medical Association Journal by scientists from Osteoporosis Canada, a nonprofit organization. Because vitamin D influences calcium absorption and may protect against osteoporosis, the authors advise an increased daily intake of 400 to 1000 IU for healthy Canadians under age 50, and up to 2000 IU for those older than 50. The researchers state the changes are necessary because winter sunlight north of the 35th parallel (which coincides with the southern border of Tennessee) provides insufficient UVB for people living in that region to adequately make vitamin D.
The studies by Knekt and Llewellyn are not the first to link vitamin D deficiency with neurological problems, however. A role for vitamin D has previously been suggested in multiple sclerosis, autism and schizophrenia.
Some experts advise interpreting the results of these and other observational studies of vitamin D with caution. The above studies relied on participants from specific geographic areas, so more study is needed to determine whether the findings apply to other regions. Furthermore, "low vitamin D levels may simply be a marker for lower health status rather than a cause of it," Andrew Grey, professor of medicine at the University of Auckland, wrote in an editorial in the Archives of Internal Medicine. This is because vitamin D levels are directly related to sunlight exposure and physical activity; less healthy individuals are therefore likely to be less active and more sunlight-deprived, and have lower levels of vitamin D.
"It is now time to test the various hypotheses generated by observational studies of vitamin D…in adequately designed and conducted randomized controlled trials," Grey concluded. "We should invest in trials that provide the best possible evidence on the benefits and risks of vitamin D before we invest in costly, difficult, and potentially unrewarding interventional strategies."
Source: Scientific America 15/7/2010
LINK TO SOURCE
_